| Change Request |
| Reference: | Change Request 293 |
| Version No: | 1.6 |
| Subject: | DSCN 57/2002 - Immunisation information for the COVER central return |
| Type of Change: | Revision of NHS data standards |
| Effective Date: | 1 January 2003 |
| Reason for Change: | Updating of immunisation data requirements |
Information about the uptake of immunisation among children aged one and two years was originally collected on DH return KC51. Similar information was also collected by CDSC, part of the Public Health Laboratory Service, through their immunisation uptake surveillance system known as COVER. Following a review of this apparent duplication the KC51 return was abolished and the DH commissioned CDSC to provide them with childhood immunisation information obtained through COVER.
The data standards supporting COVER became out of date, and the contents of this DSCN are to reflect the change in information required through COVER and to bring information requirements up to date with current national vaccination schedule.
Rubella vaccination is no longer given to adolescent girls, but given via the MMR vaccine, so RUBELLA IMMUNISATION TARGET is no longer relevant.
IMMUNISATION STATUS INDICATOR was ambiguous as it could be seen as a moving target - as a child gets older their original completed status becomes incomplete as they may require a booster. An indication of whether an immunisation primary or booster course is completed can be derived from the IMMUNISATION COMPLETION DATE, being recorded so IMMUNISATION STATUS INDICATOR was not required.
Immunisation Programmes are likely to change as new vaccines are developed, different numbers of primary doses and booster doses are given and outbreaks of disease occur or reduce. IMMUNISATION PROGRAMME has been changed to reflect this by reducing the number of examples shown and including the age group, number of primary doses and number of reinforcing doses as attributes. IMMUNISATION DISEASE TYPE has been made an attribute of IMMUNISATION PROGRAMME and VACCINATION SERVICE to comply with current NHS Data Dictionary Version.2 standards of removing any classification entity types.In addition, following Shifting the Balance of Power, immunisation target information is collected by the responsible primary care trust for a particular population. This is the population registered with the GMPs whose practice forms part of the PCT plus unregistered children resident within the PCT geographical boundary. Immunisation targets can be derived from other entity types so CHILDREN RESIDENT BY AGE GROUP was no longer required.
Summary of changes:| Name: | Barbara Fogarty |
| Date: | 28 January 2003 |
| Sponsor: | Data Standards Team |
A type of STRUCTURED PROGRAMME.
An IMMUNISATION PROGRAMMEaimed at maintaining an adequate level of immunisation in a population against a specific IMMUNISATION DISEASE TYPE by a determined sequence of IMMUNISATION COURSE TYPES. An
Each programme has a recommended number of doses which depend on the IMMUNISATION DISEASE TYPEbeing targeted. For example, Each programme has a recommended number of doses which depend on the
| Disease | Primary course doses | Reinforcing (booster) course doses | |
| Diphtheria | 3 | 2 | |
| Pertussis | 3 | 1 | |
| Tuberculosis | 1 |
| Context | Alias |
|---|---|
| plural | IMMUNISATION PROGRAMMES |
Attributes of this Class are:
| IMMUNISATION AGE GROUP | ||
| IMMUNISATION DISEASE | ||
| O | IMMUNISATION PROGRAMME TYPE | |
| applies to programmes for tuberculosis only | ||
| PRIMARY COURSE DOSE NUMBER | ||
| O | REINFORCING COURSE DOSE NUMBER |
Each IMMUNISATION PROGRAMME
| may be related to one or more RUBELLA IMMUNISATION TARGET |
Attributes of this Class are:
| O | IMMUNISATION COMPLETION DATE | |
| O | REASON FOR NO START OR COMPLETION | |
| START DATE |
Each PRIMARY CARE TRUST
| may be the receiver of one or more COMMUNITY PHARMACY SITE APPLICATION | |
| may be related to one or more DRUG MISUSE AGENCY | |
| may be related to one or more DRUG MISUSER | |
| may be related to one or more GEOGRAPHIC AREA | |
| may be in receipt of one or more GMP CLAIM FOR PAYMENT OR REIMBURSEMENT | |
| may be approver of one or more GMP CONTRACT | |
| may be the maker of one or more GMP PAYMENT OR REIMBURSEMENT | |
| may be the approver of one or more GMP TRAINING CONTRACT | |
| may be the lead PCT for one or more HEALTH PROGRAMME | |
| may be the sender of one or more PRIOR NOTIFICATION LIST FOR CYTOLOGY | |
| may be the lead PCT coordinator of one or more SMOKING CESSATION SERVICE |
A TEST OF IMMUNITYperformed as part of an IMMUNISATION PROGRAMME FOR PERSON for an IMMUNISATION DISEASE TYPE. A
| Context | Alias |
|---|---|
| plural | TESTS OF IMMUNITIES |
A type of ITEM OF SERVICE DELIVERY.
A vaccination or immunisation given to a PERSON in accordance with public policy by or on behalf of a GENERAL MEDICAL PRACTITIONER, for which a fee may be claimed.
The applicable payment rate for the VACCINATION SERVICEdelivered is determined by the combination of IMMUNISATION DISEASE TYPE and IMMUNISATION COURSE TYPE. The applicable payment rate for the
Note that ITEMS OF SERVICE DELIVERY reimbursement cannot be claimed by those
References:
Statement of Fees and Allowances Payable to General Medical Practitioners in England and Wales.
| Context | Alias |
|---|---|
| plural | VACCINATION SERVICES |
Attributes of this Class are:
| BATCH NUMBER | ||
| IMMUNISATION DISEASE | ||
| NATURE OF RISK AREA CODE | ||
| RATE OF GMP PAYMENT | ||
| VACCINATION REASON INDICATOR |
Each VACCINATION SERVICE
| K | must be classified by one and only one IMMUNISATION COURSE TYPE |
The age groups at which the immunisation status of district residents is to be recorded. The age groups at which an IMMUNISATION PROGRAMME is aimed.
Classification:
| a. | Completing primary course up to age 1 year |
| b. | Completing primary course up to age 2 years |
| c. | Completing primary course or receiving a booster by the age of 5 years |
| d. | Elderly Adult |
| e. | Other |
| Context | Alias |
|---|---|
| plural | IMMUNISATION AGE GROUPS |
The date that a course of immunisation was completed. For example, the date by which all 3 primary course doses have been given for a group C meningococcal disease vaccine.
| Context | Alias |
|---|---|
| plural | IMMUNISATION COMPLETION DATES |
A classification of IMMUNISATION DISEASE TYPE. A disease, or group of diseases, against which an IMMUNISATION PROGRAMME is directed, or for which a GENERAL MEDICAL PRACTITIONER may deliver a VACCINATION SERVICE, for which a fee may be claimed.
Examples:
| a. | Diphtheria |
| b. | Tetanus |
| c. | Poliomyelitis |
| d. | Pertussis |
| e. | Tuberculosis* |
| f. | Anthrax |
| g. | Haemophilus influenzae type b (Hib)* |
| h. | Measles, Mumps and Rubella (MMR)* |
| i. | Rabies |
| j. | Typhoid |
| k. | Group C meningococcal disease (MenC)* |
Note: This list is an example only and may be adjusted to meet local requirements.
*required to enable data to be returned centrally
References:
Statement of Fees and Allowances Payable to General Medical Practitioners in England and Wales.
| Context | Alias |
|---|---|
| plural | IMMUNISATION DISEASES |
An indicator for the result of an immunity test, showing whether a PERSON is immune or not.
Classification:
| a. | Test given and result not known | ||
| b. | Test given and result known: | ||
| i. | individual immune. For tuberculosis skin tests of immunity 'found positive' means that the individual is considered to be immune and does not require vaccination and is shown by: | ||
| | |||
| - | Mantoux test - when raised area of induration is at least 5mm in diameter, | ||
| - | Heaf test grade 2 (with complete ring or induration) or higher, or | ||
| - | Heaf test grade 1 if individual has been previously vaccinated | ||
| ii. | individual not immune. For tuberculosis skin tests of immunity 'found negative' includes Heaf grade 1 if individual has not been previously vaccinated and is considered to indicate that the individual is not immune | ||
| Context | Alias |
|---|---|
| plural | IMMUNITY TEST RESULTS |
The number of recommended doses required to complete a primary course for a particular IMMUNISATION PROGRAMME for an IMMUNISATION DISEASE. For example, for a primary course of immunisation against Diphtheria, it is recommended that 3 doses be given. The number of doses recommended may vary depending on the age of the PERSON. For example, Haemophilus influenzae type b (Hib) primary course consists of 3 doses before thirteen months or one dose thereafter and immunisation against Group C meningococcal disease (MenC) requires 2 or 3 doses before 12 months according to age at vaccination or one dose thereafter.
| Context | Alias |
|---|---|
| plural | PRIMARY COURSE DOSE NUMBER |
The number of doses required to complete a reinforcing or booster course for a particular IMMUNISATION PROGRAMME for an IMMUNISATION DISEASE. For example, for a reinforcing course of immunisation against Diphtheria, it is recommended that 2 doses be given routinely. Further reinforcing doses may be required at 10 yearly intervals for travellers to diphtheria endemic regions but these would be part of a new reinforcing or booster course.
| Context | Alias |
|---|---|
| plural | REINFORCING COURSE DOSE NUMBER |
| Central Return Form Guidance |
The Department of Health requires annual information on childhood immunisations to support performance indicators and benchmark indicators.
The performance indicators and benchmark indicators will be published routinely on the DH web site.
Information provided by COVER together with supplementary data collected on KC50 is published annually in the DH statistical bulletin: NHS Immunisation Statistics, England.
The return is required from Primary Care Trusts for children in their responsible population, i.e.
Children resident within the Primary Care Trust geographical area, who are registered with a GP belonging to another Primary Care Trust, should be returned by the GP's Primary Care Trust.
The return is required to be submitted quarterly to the Communicable Disease Surveillance Centre, who then forward annual data to the Department of Health.
The information necessary for COVER may be submitted as a computer output page containing the relevant data, which should be returned within two months of the end of the quarter to which it relates.
The COVER data provides the immunisation status of three cohorts of children, aged 12 months, 24 months, and 5 years.
This is the total number of children in the 12 month cohort, i.e. the number of children within the PRIMARY CARE TRUST's responsible population at the REPORTING PERIOD END DATE who reached the age of one during the REPORTING PERIOD.
This is a count of the number of IMMUNISATION PROGRAMME FOR PERSON for children in the 12 month cohort, with an IMMUNISATION COMPLETION DATE for an IMMUNISATION COURSE of primary up to the child's first birthday for particular IMMUNISATION DISEASES. The IMMUNISATION DISEASES currently reported are Diphtheria, Pertussis, Tetanus, Polio, Haemophilus influenzae type b (Hib), and Group C meningococcal disease (MenC).
This is the total number of children in the 24 month cohort, i.e. the number of children within the
This is a count of the number of
This is the total number of children in the 5 year cohort, i.e. the number of children within the
For primary courses this is a count of the number of
For booster courses this is a count of the number of
| Central Return Form Guidance |
The Department of Health requires annual information on childhood immunisations to support performance indicators and benchmark indicators.
The performance indicators and benchmark indicators will be published routinely on the DH web site.
Information provided by COVER together with supplementary data collected on KC50 is published annually in the DH statistical bulletin: NHS Immunisation Statistics, England.
The return is required from Primary Care Trusts for children in their responsible population, i.e.
Children resident within the Primary Care Trust geographical area, who are registered with a GP belonging to another Primary Care Trust, should be returned by the GP's Primary Care Trust.
The return is required to be submitted quarterly to the Communicable Disease Surveillance Centre, who then forward annual data to the Department of Health.
The information necessary for COVER may be submitted as a computer output page containing the relevant data, which should be returned within two months of the end of the quarter to which it relates.
The COVER data provides the immunisation status of three cohorts of children, aged 12 months, 24 months, and 5 years.
This is the total number of children in the 12 month cohort, i.e. the number of children within the PRIMARY CARE TRUST's responsible population at the REPORTING PERIOD END DATE who reached the age of one during the REPORTING PERIOD.
This is a count of the number of IMMUNISATION PROGRAMME FOR PERSON for children in the 12 month cohort, with an IMMUNISATION COMPLETION DATE for an IMMUNISATION COURSE of primary up to the child's first birthday for particular IMMUNISATION DISEASES. The IMMUNISATION DISEASES currently reported are Diphtheria, Pertussis, Tetanus, Polio, Haemophilus influenzae type b (Hib), and Group C meningococcal disease (MenC).
This is the total number of children in the 24 month cohort, i.e. the number of children within the
This is a count of the number of
This is the total number of children in the 5 year cohort, i.e. the number of children within the
For primary courses this is a count of the number of
For booster courses this is a count of the number of
| Central Return Form Guidance |
Part A of the return collects information about school leaver booster immunisations together with information on school leaver Measles, Mumps and Rubella (MMR) immunisations.
This is derived from the BIRTH DATE of the PERSON. Line 99 requires the total of children in the 13-16 age band in the period covered by the return who are included in one or more IMMUNISATION PROGRAMMES FOR PERSONS.
Column 2 requires a count of the number of children in the 13-16 age band with an IMMUNISATION DOSE GIVEN as part of an
For columns 3 and 4, the
Column 5 requires a count of the number of children in the 13-16 age band with an IMMUNISATION DOSE GIVENas part of an IMMUNISATION PROGRAMME FOR PERSONwhere the IMMUNISATION DISEASEwhich is the target of the IMMUNISATION PROGRAMMEis MMR and the IMMUNISATION COURSEis for all completed courses of MMR. The IMMUNISATION STATUS INDICATOR must indicate that the immunisation course is complete, and the IMMUNISATION COMPLETION DATE must fall within the period covered by the return, between 1 April and 31 March.
Column 5 requires a count of the number of children in the 13-16 age band with an
Part B of KC50 requires age-banded data on the number and results of tuberculosis skin tests in the year and the number of BCG vaccinations made both through
This is derived from the
Columns 7 & 8 require a count of the number of people with an IMMUNITY TEST RESULT, with a TEST OF IMMUNITY made as part of an
Column 7 requires a count of the
Column 8 requires a count of the
Note that in general the number of skin tests found negative is considerably larger than the number found positive. If this is not the case, an explanation should be included on the front sheet of the return.
The number of negative skin tests in
Columns 9 and 10 require a count of IMMUNISATION DOSES GIVEN as part of an
Column 9 requires a count of the number of people vaccinated where the IMMUNISATION PROGRAMME TYPE indicates that the
Column 10 requires a count of the number of people vaccinated where the
CM030 Community - Child Health and Immunisation for Primary Care Trusts 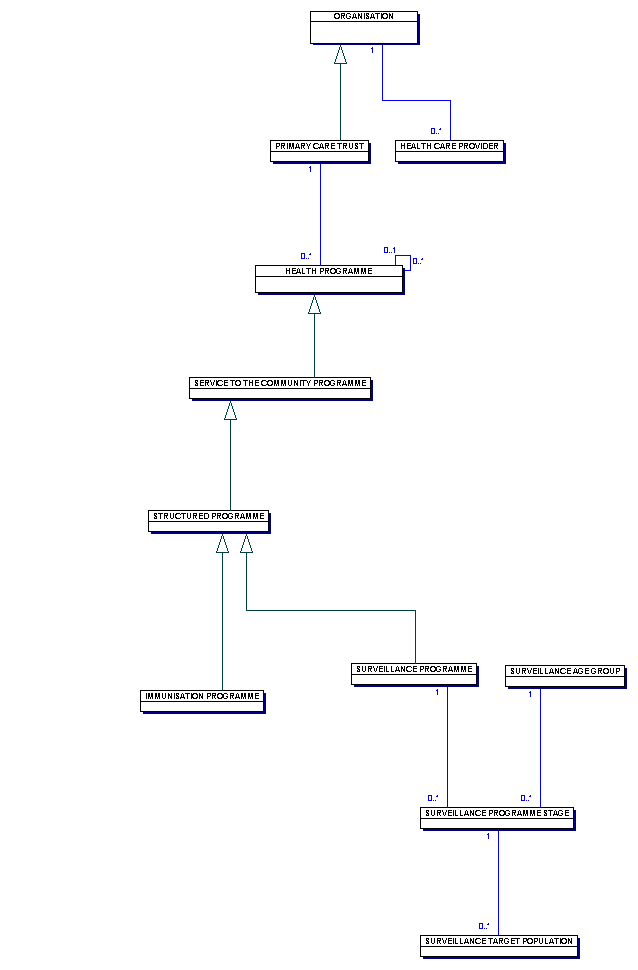
COVER Immunisation 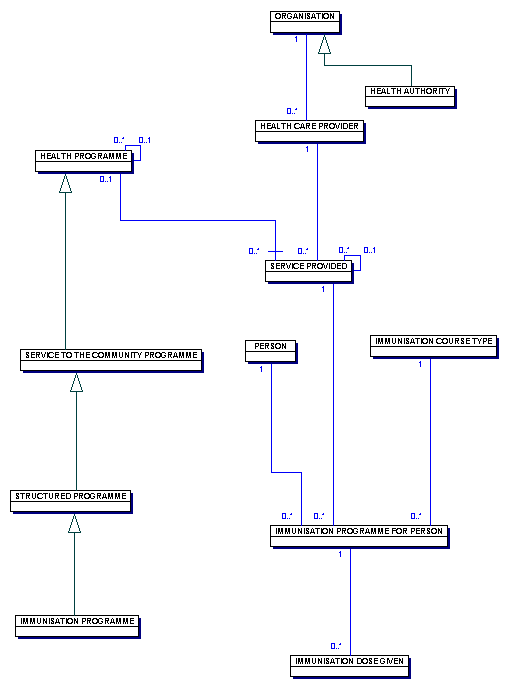
KC50 Immunisation Programmes - Activity 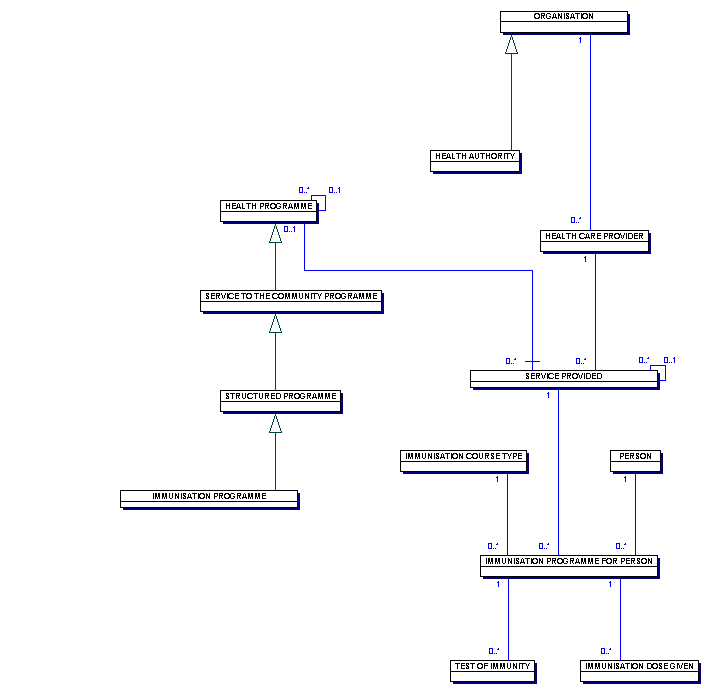
PC020 GMPS Claims and Payments/Reimbursements from Primary Care Trusts 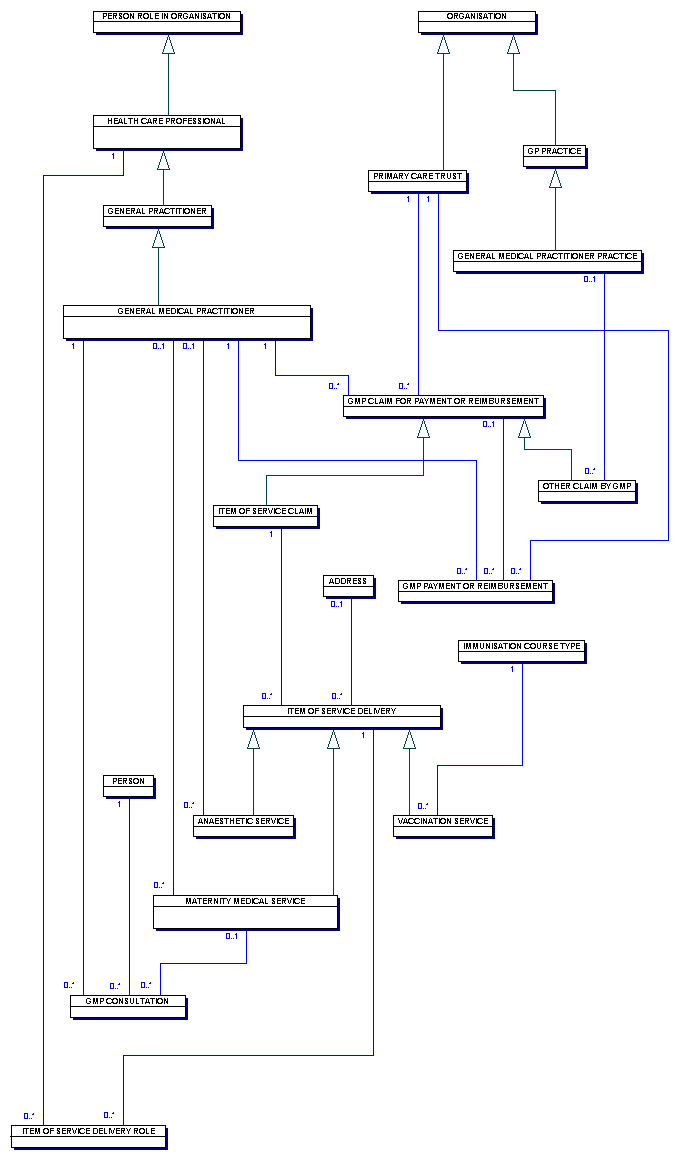
| Model View Diagram - Overview |
This diagram illustrates a PRIMARY CARE TRUSTS information requirements for two types of HEALTH PROGRAMME, immunisation and screening.
An IMMUNISATION PROGRAMME is aimed at a specific group of individuals, often within a pre-defined age band, to maintain an adequate level of immunisation for a particular disease. As an example, an IMMUNISATION PROGRAMMEfor IMMUNISATION DISEASE TYPE rubella delivers protection against the disease to one or more SURVEILLANCE PROGRAMMES.
An IMMUNISATION PROGRAMME is aimed at a specific group of individuals, often within a pre-defined age band, to maintain an adequate level of immunity against a particular disease.
For SURVEILLANCE PROGRAMMES, the broad objective is to monitor health and development. This involves an assessment of a selected group of the population by means of practical and uniform observations and measurements which can be applied rapidly.
Once a group is selected for surveillance, the type of assessment performed is often related to the age of the SURVEILLANCE AGE GROUP. There may be one or more SURVEILLANCE PROGRAMME STAGES in a
Because
PRIMARY CARE TRUSTS may contract one or more HEALTH CARE PROVIDERS to provide services within the framework of an
| Model View Diagram - Overview |
This diagram shows the classes concerned with the claims for payment which GENERAL MEDICAL PRACTITIONERS make to the PRIMARY CARE TRUST.
A
An ITEM OF SERVICE DELIVERY is delivered to a PERSON which may be recorded as delivered at a particular ADDRESS and claimed for by a particular
A HEALTH CARE PROFESSIONAL may undertake a particular role in the
The applicable payment for a particular VACCINATION SERVICEis determined by the combination of IMMUNISATION DISEASE TYPE and IMMUNISATION COURSE TYPE.
The applicable payment for a particular
A
Please address enquiries about this DSCN to:-
Data Standards Team
NHS Information Authority
Aqueous II
Aston Cross
Rocky Lane
Birmingham
B6 5RQ
Tel: 0121 333 0333